Cory:
Unlock Your AI Assistant Now!
Abstract
Background: A substantial proportion of patients with chronic coronary syndrome (CCS) undergoing percutaneous coronary intervention (PCI) experience recurrent ischaemic events. Guidelines recommend risk stratification at the time of PCI and consideration for prolonged dual antiplatelet therapy in high ischaemic risk (HIR) patients.
Aims: This study aimed to evaluate the prevalence and prognostic impact of guideline-defined HIR criteria in a large, real-world cohort of CCS patients undergoing PCI.
Methods: We retrospectively analysed CCS patients treated with drug-eluting stents at Mount Sinai Hospital between 2012 and 2022. Per the 2024 European Society of Cardiology guidelines on the management of CCS, HIR criteria included left main stenting, stent length >60 mm, diabetes mellitus, chronic kidney disease, bifurcation, and chronic total occlusion (CTO). Prognostic associations were assessed using multivariate Cox regression. The primary endpoint was major adverse cardiac and cerebrovascular events (MACCE), including death, myocardial infarction (MI), or stroke.
Results: Of 15,336 CCS patients, 10,952 (71.4%) met at least one HIR criterion. Overall, HIR status and most of its individual components, with the exception of bifurcation and CTO, predicted MACCE. Increasing numbers of HIR criteria were significantly associated with higher MACCE (p for trend<0.001), as well as the individual components of death (p for trend<0.001) and MI (p for trend<0.001).
Conclusions: In a real-world CCS cohort, guideline-defined HIR status was associated with increased MACCE. However, as almost three-quarters of patients met at least one HIR criterion, risk stratification accuracy at the time of PCI may have been limited.
Despite advancements in the management of chronic coronary syndrome (CCS), including the widespread use of drug-eluting stents (DES) and optimisation of medical therapy, a substantial proportion of patients undergoing percutaneous coronary intervention (PCI) continue to experience adverse events12. Reducing residual ischaemic risk therefore remains a key challenge in CCS management. According to the 2024 European Society of Cardiology (ESC) guidelines, a default dual antiplatelet therapy (DAPT) duration of 6 months is recommended after elective PCI3, but treatment should be adjusted based on a patient’s bleeding and ischaemic risk profile. For patients at high ischaemic risk (HIR), strategies to intensify antithrombotic therapy may include long DAPT45 or combining antiplatelet therapy with anticoagulation6. While multiple bleeding risk scores are available to guide treatment decisions7, standardised ischaemic risk assessment tools remain underdeveloped891011 and have not been systematically validated to guide decisions on prolonged antithrombotic therapy. Thrombotic risk is influenced by anatomical, procedural, and clinical factors12. As highlighted in the 2024 ESC CCS guidelines, key clinical risk factors include diabetes mellitus and chronic kidney disease (CKD); additionally, procedural characteristics that further elevate ischaemic risk include treatment of chronic total occlusions (CTO), bifurcation lesions requiring stenting of both the main and side branches, total stent length >60 mm, and left main PCI. This study aims to evaluate the prevalence and prognostic impact of such criteria from a large, real-world cohort of PCI patients.
Methods
Study population
Consecutive patients who underwent PCI at Mount Sinai Hospital (New York, NY, USA) between 2012 and 2022 were assessed for eligibility. The inclusion criteria were a CCS indication for PCI and implantation of DES. Patients with missing information on HIR criteria and patients lost to 30-day follow-up were excluded. The study adhered to the Declaration of Helsinki and received approval from the Institutional Review Board. After providing informed consent, patients’ baseline and procedural data were included in the institutional database. Dedicated research personnel were responsible for conducting follow-up at 1 year after the intervention, either through in-person visits or telephone calls. Postprocedural therapy was left to the physician’s discretion, in accordance with current guidelines. Complex PCI was defined according to established criteria as the presence of ≥1 of the following procedural features: treatment of 3 vessels or lesions, implantation of ≥3 stents, bifurcation PCI requiring 2 stents, total stent length >60 mm, or a CTO as the target lesion13.
The study population was categorised based on 6 predefined HIR criteria3: diabetes, CKD, bifurcation lesions requiring 2 or more stents, stent length >60 mm, CTO, and left main coronary artery involvement. Patients were classified as being at HIR if they met at least one of these criteria. Supplementary Table 1 shows the definition of the HIR criteria according to the 2024 ESC guidelines as compared with the present study.
Study outcomes and definitions
The primary outcome was major adverse cardiac and cerebrovascular events (MACCE), a composite of all-cause death, myocardial infarction (MI), or stroke 1 year after PCI. Secondary outcomes included the individual MACCE components, target vessel revascularisation, definite or probable stent thrombosis, post-discharge bleeding, and all bleeding. Post-discharge bleeding was defined as any bleeding event occurring after the index hospitalisation for PCI that required a blood transfusion or a repeat hospitalisation. MI was defined according to the Third Universal Definition, and only spontaneous events were considered14. All in-hospital events and post-discharge events leading to readmission at our institution were independently adjudicated by a clinical events committee.
Statistical analysis
Continuous variables are presented as mean±standard deviation or median (interquartile range) and were compared across HIR groups using the Student’s t-test. Categorical variables are reported as counts and percentages and were analysed using the χ² test with Yates’s correction for continuity or Fisher’s exact test, as appropriate. Time-to-event outcomes were assessed using the Kaplan-Meier method, with differences between groups evaluated using the log-rank test. Associations between HIR criteria and clinical outcomes were examined using both univariate and multivariate regression models. To evaluate the prognostic impact of the cumulative number of HIR criteria, Cox proportional hazards models were used, treating the number of criteria as an ordinal variable to estimate unadjusted hazard ratios (HRs) with corresponding 95% confidence intervals (CIs). An UpSet plot was generated to visualise the most frequent combinations of HIR criteria. All reported p-values are two-tailed, with p<0.05 considered significant. Statistical analysis was performed using Stata, version 18.0 (StataCorp).
Results
Baseline characteristics
A total of 15,336 CCS patients underwent PCI with DES at Mount Sinai Hospital between 2012 and 2022 and were included in the present analysis. Among these, 4,384 patients (28.58%) were classified as non-HIR, while 10,952 (71.41%) were at HIR. In particular, 6,412 (41.8%) had 1 HIR criterion, 3,522 (22.97%) had 2 criteria, and 1,018 (6.64%) had 3 or more. Diabetes was the most common risk factor, affecting 7,437 individuals (48.5%), followed by CKD (4,165; 27.16%), bifurcation lesions (1,501; 9.78%), stent length >60 mm (1,405; 9.16%), CTO (1,125; 7.33%), and left main involvement (1,030; 6.7%). Following diabetes alone, diabetes plus CKD was the most common profile of HIR features (Figure 1).
Table 1 and Table 2 show the baseline clinical and procedural characteristics, respectively, in patients with no, 1, 2, or ≥3 HIR criteria. Patients with a higher number of HIR criteria tended to be older and presented a greater prevalence of cardiovascular risk factors. Higher-risk patients also exhibited a higher prevalence of multivessel disease, B2/C lesions, and moderate to severe calcifications, as well as progressively longer lesions (Table 2). Consistently, the use of advanced calcium modification techniques, particularly rotational atherectomy, was significantly more frequent among patients with multiple HIR criteria, increasing from 13.4% in patients with no HIR criteria to 37.6% in those with ≥3 criteria (p<0.001). Intracoronary imaging was used in a minority of cases: while intravascular ultrasound utilisation was similar across risk groups, optical coherence tomography (OCT) use declined with increasing risk burden (4.8% in non-HIR vs 2.4% in ≥3 criteria HIR patients; p<0.001) (Table 2).
Prescription of statin and dual antiplatelet therapy at discharge was similar across all groups, but there was a stepwise increase of prasugrel, anticoagulant, and beta blocker use in higher-risk patients (Table 2).
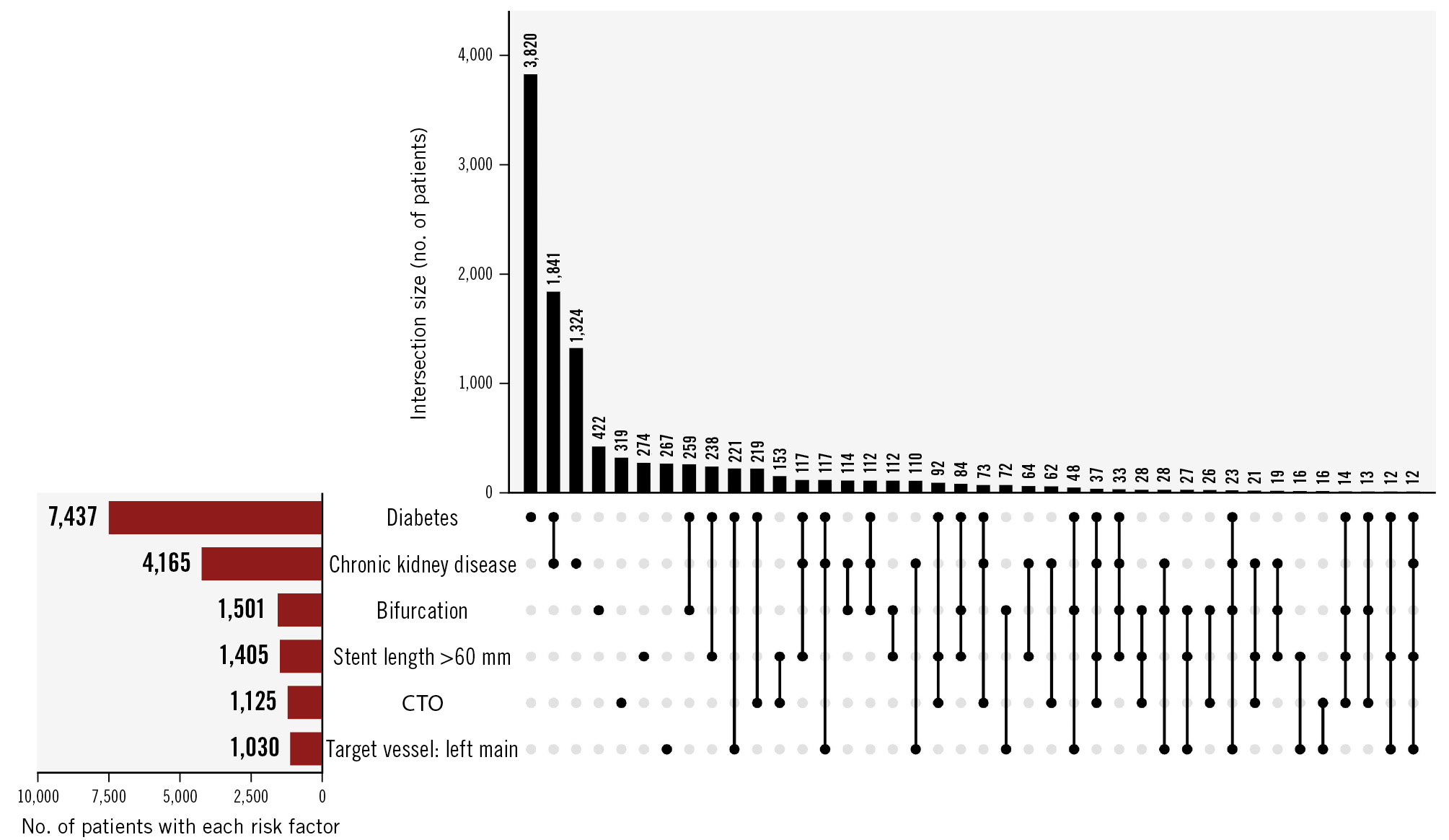
Figure 1. UpSet plot of high-risk clinical and anatomical features. The horizontal red bars represent the number of patients with each individual risk factor (diabetes, chronic kidney disease, bifurcation, stent length >60 mm, chronic total occlusion, and left main target vessel). The vertical black bars indicate the number of patients with overlapping criteria, with connected black dots denoting which factors are included in each intersection. CTO: chronic total occlusion
Table 1. Baseline clinical characteristics stratified by the number of risk criteria.
| No risk criteria N=4,384 (28.6) | 1 risk criterion N=6,412 (41.8) | 2 risk criteria N=3,522 (23.0) | ≥3 risk criteria N=1,018 (6.6) | p-value | |
|---|---|---|---|---|---|
| Age, years | 66.1±11.0 | 66.6±10.9 | 67.9±10.8 | 68.1±10.9 | <0.001 |
| BMI, kg/m2 | 28.2±5.2 | 28.9±5.5 | 28.9±5.7 | 28.9±5.6 | <0.001 |
| Female sex | 977 (22.3) | 1,710 (26.7) | 1,077 (30.6) | 272 (26.7) | <0.001 |
| Race/ethnicity | <0.001 | ||||
| Caucasian | 2,239 (51.1) | 2,477 (38.6) | 1,308 (37.1) | 445 (43.7) | |
| African-American | 344 (7.8) | 582 (9.1) | 352 (10.0) | 104 (10.2) | |
| Asian | 505 (11.5) | 1,105 (17.2) | 585 (16.6) | 192 (18.9) | |
| Hispanic | 591 (13.5) | 1,036 (16.2) | 614 (17.4) | 139 (13.7) | |
| Others | 705 (16.1) | 1,212 (18.9) | 663 (18.8) | 138 (13.6) | |
| Current smoker | 556 (12.7) | 682 (10.6) | 299 (8.5) | 95 (9.3) | <0.001 |
| Family history of CAD | 892 (20.3) | 1,173 (18.3) | 609 (17.3) | 165 (16.2) | <0.001 |
| Anaemia | 930 (21.2) | 2,299 (35.9) | 1,826 (51.8) | 542 (53.2) | <0.001 |
| Diabetes mellitus | 0 (0) | 3,810 (59.4) | 2,777 (78.8) | 850 (83.5) | N/A |
| Hypertension | 3,960 (90.3) | 6,142 (95.8) | 3,401 (96.6) | 989 (97.2) | <0.001 |
| Hyperlipidaemia | 4,007 (91.4) | 6,063 (94.6) | 3,388 (96.2) | 963 (94.6) | <0.001 |
| Lung disease | 265 (6.0) | 391 (6.1) | 235 (6.7) | 60 (5.9) | 0.612 |
| Peripheral artery disease | 276 (6.3) | 566 (8.8) | 452 (12.8) | 128 (12.6) | <0.001 |
| Cerebrovascular disease | 312 (7.1) | 677 (10.6) | 469 (13.3) | 137 (13.5) | <0.001 |
| Atrial fibrillation | 360 (8.2) | 489 (7.6) | 320 (9.1) | 92 (9.0) | 0.060 |
| Dialysis | 7 (0.2) | 124 (1.9) | 333 (9.5) | 107 (10.5) | <0.001 |
| Chronic kidney disease | 0 (0) | 1,321 (20.6) | 2,190 (62.2) | 654 (64.2) | N/A |
| Prior PCI | 1,898 (43.3) | 3,002 (46.8) | 1,774 (50.4) | 528 (51.9) | <0.001 |
| Prior MI | 959 (21.9) | 1,363 (21.3) | 878 (24.9) | 295 (29.0) | <0.001 |
| Prior CABG | 477 (10.9) | 948 (14.8) | 725 (20.6) | 251 (24.7) | <0.001 |
| LVEF, % | 56.6±9.3 | 55.9±10.0 | 54.3±11.3 | 52.5±12.3 | <0.001 |
| Data are presented as n (%) or mean±standard deviation. BMI: body mass index; CABG: coronary artery bypass graft; CAD: coronary artery disease; LVEF: left ventricular ejection fraction; MI: myocardial infarction; N/A: not available; PCI: percutaneous coronary intervention | |||||
Table 2. Baseline procedural characteristics stratified by the number of risk criteria.
| No risk criteria N=4,384 (28.6) | 1 risk criterion N=6,412 (41.8) | 2 risk criteria N=3,522 (23.0) | ≥3 risk criteria N=1,018 (6.6) | p-value | |
|---|---|---|---|---|---|
| PCI vessels | |||||
| Left main | 0 (0) | 266 (4.1) | 435 (12.4) | 329 (32.3) | N/A |
| LAD | 2,780 (63.4) | 3,981 (62.1) | 2,152 (61.1) | 682 (67.0) | 0.003 |
| LCx | 1,783 (40.7) | 2,911 (45.4) | 1,719 (48.8) | 586 (57.6) | <0.001 |
| RCA | 1,844 (42.1) | 2,820 (44.0) | 1,682 (47.8) | 491 (48.2) | <0.001 |
| SVG | 85 (1.9) | 152 (2.4) | 130 (3.7) | 33 (3.2) | <0.001 |
| Multivessel disease | 2,429 (55.4) | 4,043 (63.1) | 2,523 (71.6) | 848 (83.3) | <0.001 |
| Lesion profile | |||||
| Moderate to severe calcification | 821 (18.7) | 1,350 (21.1) | 962 (27.3) | 444 (43.6) | <0.001 |
| Bifurcation lesions | 609 (13.9) | 1,084 (16.9) | 842 (23.9) | 523 (51.4) | <0.001 |
| Chronic total occlusion | 0 (0) | 319 (5.0) | 476 (13.5) | 330 (32.4) | N/A |
| Total stent length, mm | 26.6±12.3 | 30.8±17.7 | 40.4±24.8 | 66.4±27.0 | <0.001 |
| No. of stents implanted | 1.3±0.5 | 1.4±0.6 | 1.6±0.8 | 2.4±0.9 | <0.001 |
| Max. stent diameter, mm | 3.1±0.5 | 3.1±0.5 | 3.1±0.5 | 3.3±0.5 | <0.001 |
| Preprocedural TIMI flow | 2.9±0.4 | 2.8±0.7 | 2.6±1.0 | 2.1±1.3 | <0.001 |
| Postprocedural TIMI flow | 3.0±0.1 | 3.0±0.2 | 3.0±0.3 | 2.9±0.4 | <0.001 |
| SYNTAX score | 10.5±7.9 | 12.3±9.1 | 16.0±11.0 | 24.7±13.3 | <0.001 |
| Complex PCI* | 713 (16.3) | 2,092 (32.6) | 1,928 (54.7) | 964 (94.7) | <0.001 |
| Access site | |||||
| Radial access | 1,231 (28.1) | 1,469 (22.9) | 640 (18.1) | 125 (12.3) | <0.001 |
| Femoral access | 3,089 (70.5) | 4,862 (75.8) | 2,826 (80.2) | 871 (85.6) | <0.001 |
| Calcium modification techniques | |||||
| Rotational atherectomy | 588 (13.4) | 1,022 (15.9) | 807 (22.9) | 383 (37.6) | <0.001 |
| Orbital atherectomy | 171 (3.9) | 199 (3.1) | 111 (3.2) | 32 (3.1) | 0.118 |
| ELCA | 14 (0.3) | 23 (0.4) | 24 (0.7) | 8 (0.8) | 0.020 |
| Intracoronary imaging | |||||
| OCT | 210 (4.8) | 253 (4.0) | 96 (2.7) | 24 (2.4) | <0.001 |
| IVUS | 374 (8.5) | 530 (8.3) | 301 (8.6) | 101 (9.9) | 0.376 |
| Discharge medication | |||||
| DAPT | 4,084 (93.2) | 6,021 (93.9) | 3,275 (93.0) | 944 (92.7) | 0.110 |
| Aspirin | 4,099 (93.5) | 6,032 (94.1) | 3,292 (93.5) | 947 (93.0) | 0.211 |
| P2Y12 inhibitor | 4,350 (99.2) | 6,381 (99.5) | 3,489 (99.1) | 1,006 (98.8) | 0.006 |
| Prasugrel | 444 (10.1) | 686 (10.7) | 411 (11.7) | 145 (14.2) | <0.001 |
| Ticagrelor | 606 (13.8) | 941 (14.7) | 497 (14.1) | 167 (16.4) | 0.082 |
| Anticoagulant | 320 (7.3) | 466 (7.3) | 309 (8.8) | 105 (10.3) | <0.001 |
| Statin | 4,181 (95.4) | 6,077 (94.8) | 3,355 (95.3) | 971 (95.4) | 0.166 |
| Beta blocker | 3,257 (74.3) | 5,009 (78.1) | 2,929 (83.2) | 884 (86.8) | <0.001 |
| Data are presented as n (%) or mean±standard deviation. *Complex PCI was defined according to established criteria as the presence of one or more of the following: treatment of 3 vessels or lesions, implantation of 3 or more stents, bifurcation PCI requiring 2 stents, total stent length >60 mm, or a CTO as the target lesion. CTO: chronic total occlusion; DAPT: dual antiplatelet therapy; ELCA: excimer laser coronary atherectomy; IVUS: intravascular ultrasound; LAD: left anterior descending artery; LCx: left circumflex artery; N/A: not available; OCT: optical coherence tomography; PCI: percutaneous coronary intervention; RCA: right coronary artery; SVG: saphenous vein graft; TIMI: Thrombolysis in Myocardial Infarction | |||||
Clinical outcomes at 1 year
A statistically significant increase in MACCE risk was observed with a higher number of HIR criteria: 1.9% with none, 2.9% with 1, 6.1% with 2, and 8.4% with 3 or more (p-trend<0.001) (Figure 2). Similar trends were noted for all-cause death, MI, and bleeding (Table 3). As compared with patients having no HIR criteria, the risk of experiencing a MACCE was significantly increased in patients with 1 criterion (HR 1.58, 95% CI: 1.19-2.09), 2 criteria (HR 3.28, 95% CI: 2.48-4.33), and 3 or more criteria (HR 4.73, 95% CI: 3.40-6.58) (Table 3, Central illustration). Patients with greater risk burden factors also exhibited a markedly increased mortality risk (p for trend<0.001), with an HR of 6.55 (95% CI: 4.12-10.40) in the ≥3 risk criteria group, an HR of 4.14 (95% CI: 2.75-6.23) in the 2 risk criteria group, and an HR of 1.70 (95% CI: 1.11-2.59) in the 1 risk criterion group. Similarly, across the 4 risk groups, there was a stepwise increase in the risks of MI and overall bleeding, the latter mainly driven by periprocedural complications (Table 3, Central illustration).
In the multivariate regression analysis, CKD was the strongest predictor of MACCE (HR 2.69, 95% CI: 2.17-3.33), followed by left main PCI (HR 2.00, 95% CI: 1.41-2.84), diabetes (HR 1.69, 95% CI: 1.35-2.11), and stent length >60 mm (HR 1.45, 95% CI: 1.08-1.94) (Table 4, Central illustration).
In a supplementary multivariate model further adjusted for intracoronary imaging use, imaging was independently associated with a lower risk of MACCE (HR 0.59, 95% CI: 0.39-0.89; p=0.012), while the prognostic impact of individual ESC HIR criteria remained largely unchanged (Supplementary Table 2).
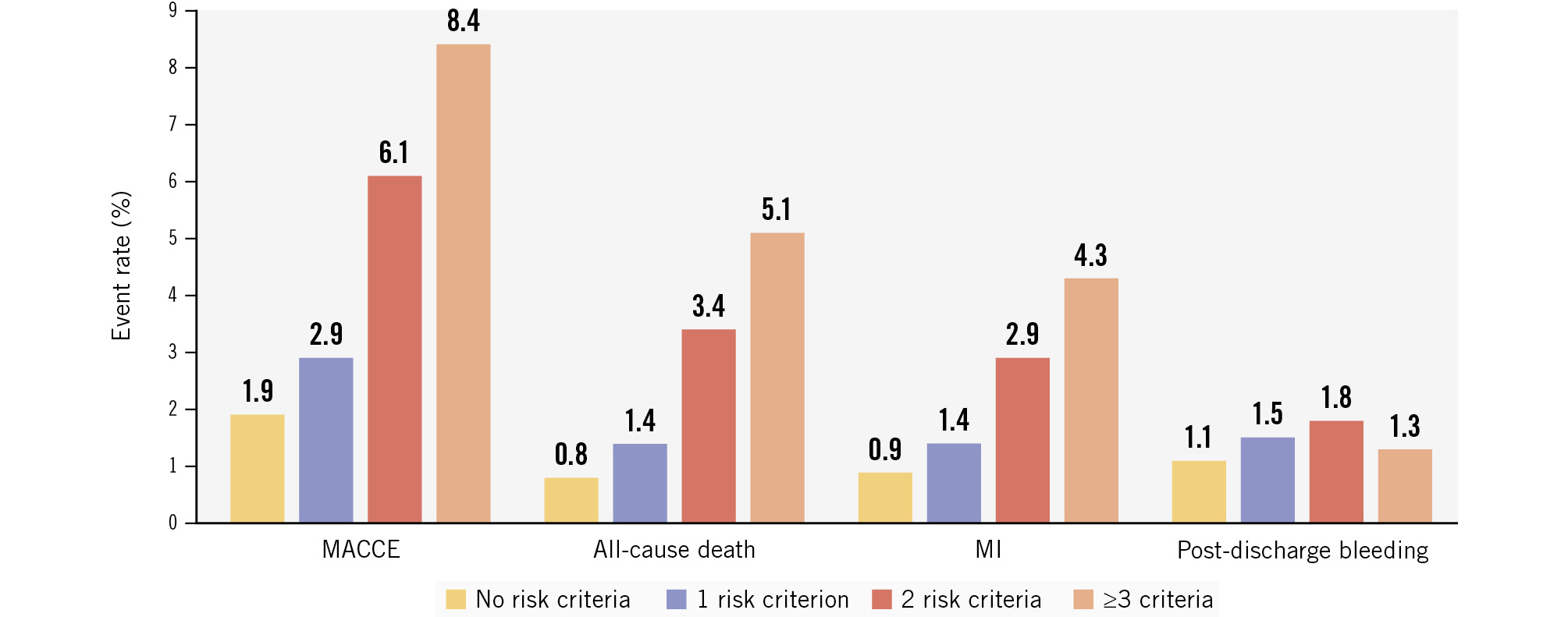
Figure 2. One-year adverse event rates stratified by risk group. Bar chart comparing the incidence of MACCE, all-cause death, MI, and post-discharge bleeding across the 4 treatment groups (no risk criteria, 1 risk criterion, 2 risk criteria, and ≥3 risk criteria). MACCE: major adverse cardiac and cerebrovascular events; MI: myocardial infarction
Table 3. Association between the number of HIR criteria and adverse events at one year.
| No. of patients | No. of events | HR (95% CI) | p-value | Trend p-value | |
|---|---|---|---|---|---|
| MACCE (death, MI, or stroke) | |||||
| No risk criteria | 4,384 | 69 (1.9) | Ref. | <0.001 | |
| 1 risk criterion | 6,412 | 158 (2.9) | 1.58 (1.19-2.09) | 0.002 | |
| 2 risk criteria | 3,522 | 177 (6.1) | 3.28 (2.48-4.33) | <0.001 | |
| ≥3 risk criteria | 1,018 | 72 (8.4) | 4.73 (3.40-6.58) | <0.001 | |
| All-cause death | |||||
| No risk criteria | 4,384 | 30 (0.8) | Ref. | <0.001 | |
| 1 risk criterion | 6,412 | 74 (1.4) | 1.70 (1.11-2.59) | 0.015 | |
| 2 risk criteria | 3,522 | 98 (3.4) | 4.14 (2.75-6.23) | <0.001 | |
| ≥3 risk criteria | 1,018 | 44 (5.1) | 6.55 (4.12-10.40) | <0.001 | |
| MI | |||||
| No risk criteria | 4,384 | 35 (0.9) | Ref. | <0.001 | |
| 1 risk criterion | 6,412 | 75 (1.4) | 1.48 (0.99-2.20) | 0.058 | |
| 2 risk criteria | 3,522 | 83 (2.9) | 3.03 (2.00-4.49) | <0.001 | |
| ≥3 risk criteria | 1,018 | 36 (4.3) | 4.65 (2.92-7.41) | <0.001 | |
| Stroke | |||||
| No risk criteria | 4,384 | 7 (0.2) | Ref. | 0.370 | |
| 1 risk criterion | 6,412 | 16 (0.3) | 1.57 (0.65-3.83) | 0.317 | |
| 2 risk criteria | 3,522 | 10 (0.3) | 1.82 (0.69-4.77) | 0.226 | |
| ≥3 risk criteria | 1,018 | 2 (0.2) | 1.28 (0.27-6.16) | 0.759 | |
| Post-discharge bleeding | |||||
| No risk criteria | 4,384 | 42 (1.1) | Ref. | 0.062 | |
| 1 risk criterion | 6,412 | 84 (1.5) | 1.38 (0.95-1.99) | 0.090 | |
| 2 risk criteria | 3,522 | 54 (1.8) | 1.63 (1.08-2.43) | 0.018 | |
| ≥3 risk criteria | 1,018 | 12 (1.3) | 1.27 (0.67-2.41) | 0.466 | |
| All bleeding | |||||
| No risk criteria | 4,384 | 99 (2.4) | Ref. | <0.001 | |
| 1 risk criterion | 6,412 | 203 (3.3) | 1.41 (1.11-1.79) | 0.005 | |
| 2 risk criteria | 3,522 | 143 (4.3) | 1.81 (1.40-2.34) | <0.001 | |
| ≥3 risk criteria | 1,018 | 54 (5.4) | 2.40 (1.72-3.34) | <0.001 | |
| Data are presented as n (%) unless otherwise indicated. CI: confidence interval; HIR: high ischaemic risk; HR: hazard ratio; MACCE: major adverse cardiac and cerebrovascular events; MI: myocardial infarction | |||||
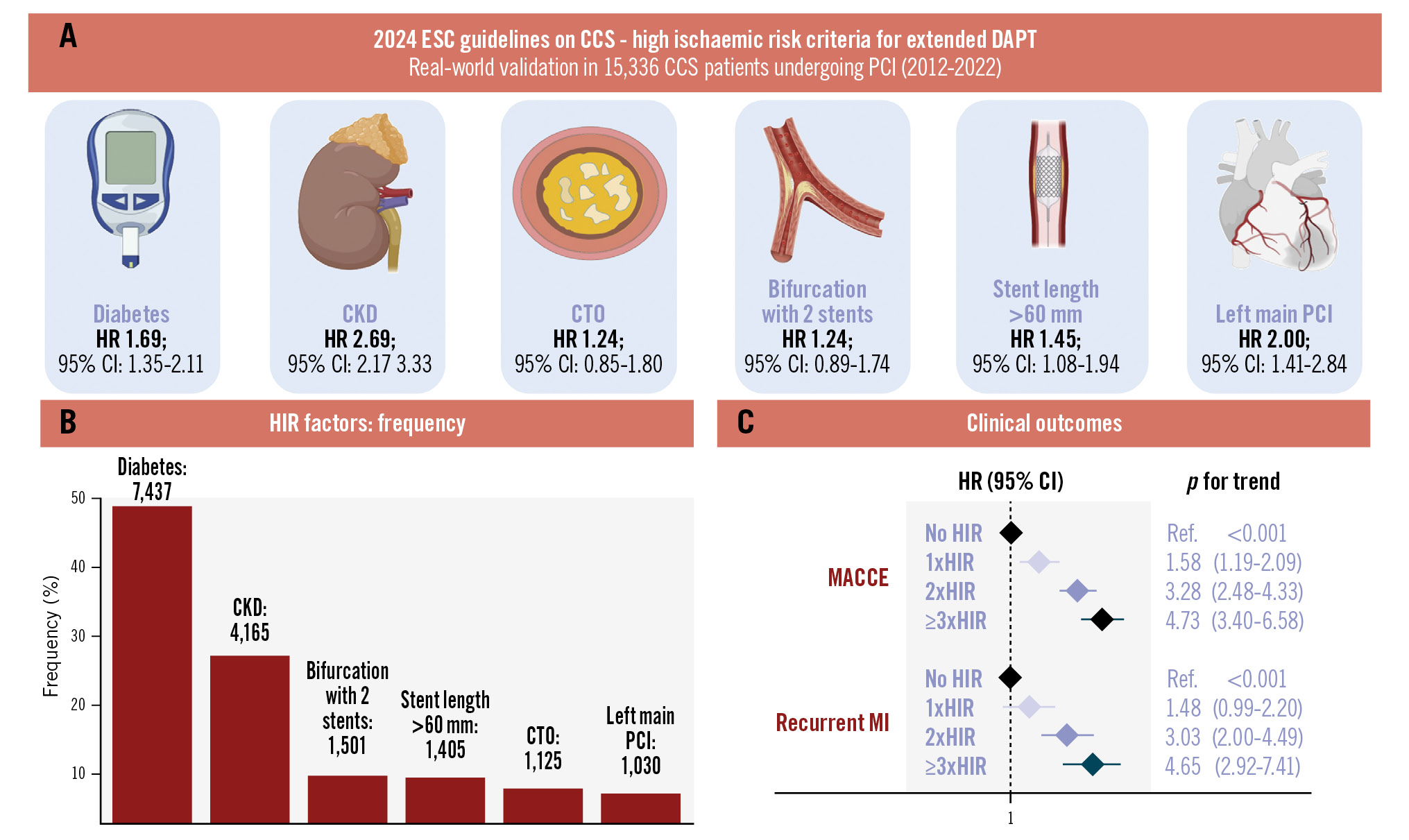
Central illustration. High ischaemic risk criteria identify PCI patients with stepwise increases in adverse events. The number of HIR features is associated with a stepwise increase in ischaemic events after PCI. A) Multivariate-adjusted hazard ratios for individual HIR criteria. B) Frequency of HIR factors in the study population. C) Forest plot of MACCE and MI outcomes across the 4 risk groups. CCS: chronic coronary syndrome; CI: confidence interval; CKD: chronic kidney disease; CTO: chronic total occlusion; DAPT: dual antiplatelet therapy; ESC: European Society of Cardiology; HIR: high ischaemic risk; HR: hazard ratio; MACCE: major adverse cardiac and cerebrovascular events; MI: myocardial infarction; PCI: percutaneous coronary intervention
Table 4. Univariate and multivariate model for MACCE at one year after the index procedure.
| Univariate HR (95% CI) | p-value | Multivariate HR (95% CI) | p-value | |
|---|---|---|---|---|
| ESC risk criteria | ||||
| Diabetes | 1.71 (1.42-2.06) | <0.001 | 1.69 (1.35-2.11) | <0.001 |
| CKD | 2.78 (2.32-3.33) | <0.001 | 2.69 (2.17-3.33) | <0.001 |
| CTO | 1.04 (0.74-1.46) | 0.827 | 1.24 (0.85-1.80) | 0.273 |
| Bifurcation | 1.35 (1.03-1.77) | 0.028 | 1.24 (0.89-1.74) | 0.208 |
| Stent length >60 mm | 1.53 (1.16-2.00) | 0.002 | 1.45 (1.08-1.94) | 0.012 |
| Left main PCI | 2.19 (1.68-2.86) | <0.001 | 2.00 (1.41-2.84) | <0.001 |
| CI: confidence interval; CKD: chronic kidney disease; CTO: chronic total occlusion; ESC: European Society of Cardiology; HR: hazard ratio; MACCE: major adverse cardiac and cerebrovascular events; PCI: percutaneous coronary intervention | ||||
Discussion
This study represents the first large-scale, real-world validation of the high ischaemic risk criteria proposed by the recent 2024 European Society of Cardiology guidelines for patients with chronic coronary syndrome undergoing PCI with DES. The main findings of this study can be summarised as follows: (i) HIR criteria are common in patients undergoing PCI, and their presence is associated with an increased risk of ischaemic events, with the additive risk effect conferred by multiple criteria; (ii) diabetes, chronic kidney disease, left main involvement, and longer total stent length are strongly associated with adverse outcomes; and (iii) HIR criteria also increase the risk of bleeding, representing a challenge for the adequate balance between ischaemic and bleeding risks.
Our study supports the prognostic value of several well-established clinical and anatomical risk factors, including diabetes mellitus, CKD, left main involvement, and greater total stent length − all independently associated with adverse ischaemic outcomes15161718. The ischaemic event rates in this population were consistent with those reported in prior observational registries and clinical trials, and the high prevalence of HIR reflects the broad inclusivity of these criteria in current clinical practice1920. In our cohort of patients, MACCE rates increased from 1.9% in patients with no risk factors to 8.4% in those with 3 or more risk factors, reflecting the cumulative risk pattern observed in other populations with atherosclerotic cardiovascular disease21. For example, in the REACH registry, including COMPASS-eligible patients with stable vascular disease, each additional enrichment criterion (e.g., diabetes, renal impairment, peripheral artery disease) was linked to a stepwise increase in 4-year MACCE risk, reaching a 4-fold rise with ≥4 criteria22.
Interestingly, bifurcation with 2 stents and CTO PCI were not significantly associated with 1-year MACCE in this study, despite both being historically regarded as HIR features. This can be explained by the substantial improvements in current PCI practice, including the use of second-generation DES; procedural refinements, such as proximal optimisation technique and final kissing balloon inflation; and a better selection of stenting strategies232425. In addition, the integration of intracoronary imaging has contributed to improved bifurcation PCI outcomes as demonstrated in a recent randomised trial, in which routine OCT-guided PCI in complex bifurcations reduced 2-year MACCE compared with angiography guidance (10.1% vs 14.1%; HR 0.70)26. Indeed, intravascular imaging may help mitigate procedural contributors to both ischaemic and bleeding complications by improving stent optimisation and allowing early detection of complications such as malapposition or edge dissection, aspects particularly relevant in patients with multiple HIR criteria27.
In the present study, although the overall use of intracoronary imaging was relatively low, reflecting contemporary practice patterns, its use was independently associated with a significant reduction in 1-year MACCE after adjustment for ESC-defined HIR features. This finding underscores the potential clinical impact of imaging-guided PCI in patients at HIR and suggests that broader adoption of intracoronary imaging may represent an actionable strategy to reduce residual risk in routine clinical practice.
Similarly, CTO PCI success rates have improved because of advancements in devices, techniques, and operator expertise. In the Japanese CTO-PCI Expert Registry of nearly 4,000 cases, 1-year target lesion revascularisation and non-fatal MI rates were only 4.8% and 0.2%, respectively28. Also, while bifurcation lesions were not associated with MACCE in multivariate analysis, they did show an association in the univariate model, suggesting that their risk is largely explained by coexisting stronger predictors.
Our study further supports the ESC 2024 HIR framework as a useful risk stratification tool with practical implications for tailoring antithrombotic therapy. However, the findings that nearly three-quarters of PCI patients meet ≥1 HIR criterion and that HIR criteria were associated with increased bleeding have important implications for real-world practice. Indeed, in our cohort, overall bleeding complications occurred in 5.4% of patients with 3 or more risk factors, mainly driven by periprocedural complications.
Prior studies have shown that major and late bleeding confer a mortality risk comparable to MI, with early bleeding after PCI having an even stronger association29. The overlap of ischaemic and bleeding risk factors, including age, diabetes, and CKD, highlights the need to balance both risks carefully when selecting treatment strategies. Evidence from a large pooled analysis of randomised trials suggests that the ischaemic benefit of prolonged DAPT is primarily observed in patients without high bleeding risk, regardless of procedural complexity, emphasising the need to prioritise bleeding risk when individualising antithrombotic strategies30. Novel antithrombotic strategies may offer a better balance between ischaemic and bleeding risks, including the use of ticagrelor monotherapy, which, in a post hoc analysis of the TWILIGHT trial, was not associated with excess ischaemic or bleeding events regardless of HIR status31.
Limitations
Our study should be interpreted in light of some limitations. First, we considered all-cause mortality and not cardiovascular mortality in the assessment of the primary outcome – a less specific but more patient-oriented outcome. Given the difficulties in establishing the cause of death, using all-cause mortality is less likely to introduce bias and is in keeping with recommendations from the Academic Research Consortium-2 consensus32. Second, detailed information on dual antiplatelet therapy duration at hospital discharge was not available. Third, some of the least common HIR criteria specified in current guidelines were not collected in our population, which may have limited our ability to fully capture ischaemic risk. These include a history of recurrent MI, a history of stent thrombosis occurring while on antiplatelet therapy, stenting of a less patent vessel, treatment of at least 3 lesions, implantation of 3 or more stents, concomitant inflammatory and/or prothrombotic disease, premature or accelerated coronary artery disease (CAD), multivessel CAD, polyvascular disease, and intravascular imaging-assessed stent underexpansion. Fourth, data on invasive coronary physiology were not systematically collected in this retrospective cohort and could not be analysed. Fifth, the prevalence of intravascular imaging use and radial access for PCI were relatively low, which is in line with contemporary data from US nationwide studies333435. Lastly, the single-centre cohort design limits generalisability and may restrict the applicability of our findings in broader, more diverse clinical settings.
Conclusions
In this large contemporary cohort of CCS patients undergoing PCI, the ESC 2024 HIR criteria effectively identified patients at increased risk of ischaemic events at 1 year, with a significant stepwise relationship between an escalating number of criteria and incrementally adverse outcomes. Our findings confirm the prognostic relevance of several clinical and anatomical factors, particularly diabetes, CKD, left main disease, and long stent length, while also highlighting the limited predictive value of certain procedural features, such as bifurcation and CTO interventions, in the current era of optimised PCI techniques. Given the high prevalence of HIR features in real-world practice and the partial overlap with bleeding risk factors, while the HIR framework represents a valuable tool for risk stratification and treatment tailoring, future efforts should aim to refine this classification into a more granular, personalised system to better predict ischaemic risk.
Impact on daily practice
The European Society of Cardiology 2024 high ischaemic risk (HIR) criteria identify percutaneous coronary intervention (PCI) patients at increased risk of ischaemic events, particularly those with diabetes, chronic kidney disease, left main involvement, or longer total stent length. Bifurcation lesion and chronic total occlusion PCI have limited prognostic value in the contemporary drug-eluting stent era. These findings support the use of the HIR framework to guide individualised antithrombotic therapy while carefully balancing ischaemic and bleeding risks in real-world practice.
Conflict of interest statement
R. Mehran reports institutional research grants from Abbott, Abiomed, Applied Therapeutics, AstraZeneca, Bayer, Beth Israel Deaconess, Bristol-Myers Squibb, CERC, Chiesi, Concept Medical, CSL Behring, DSI, Medtronic, Novartis Pharmaceuticals, and OrbusNeich; consultant fees from Abbott, Boston Scientific, Janssen Scientific Affairs, Medscape/WebMD, Medtelligence (Janssen Scientific Affairs), Roivant Sciences, Sanofi, and Siemens Medical Solutions; consultant fees paid to the institution from Abbott and Bristol-Myers Squibb; advisory board, funding paid to the institution from Spectranetics/Philips/Volcano; consultant fees (spouse) from Abiomed, The Medicines Company, and Merck; equity <1% from Claret Medical and Elixir Medical; DSMB membership fees paid to the institution from Watermark Research Partners; consulting (no fee) for Idorsia Pharmaceuticals Ltd. and Regeneron Pharmaceuticals; and Associate Editor for ACC and AMA. The other authors have no conflicts of interest to declare.
Supplementary data
To read the full content of this article, please download the PDF.

