Cory:
Unlock Your AI Assistant Now!
Abstract
Background: Complex percutaneous coronary intervention (PCI) is increasingly performed among patients undergoing myocardial revascularisation.
Aims: We conducted a systematic review and meta-analysis to evaluate the association between complex PCI and the risk of ischaemic and bleeding outcomes.
Methods: Hazard ratios (HRs) were pooled using a random-effects model within a Bayesian framework. The primary analysis was restricted to studies providing adjusted risk estimates, whereas the secondary analysis included unadjusted risk estimates. The primary outcomes were myocardial infarction and major bleeding. The secondary outcomes were all-cause death, stent thrombosis, cardiovascular death, target lesion or vessel revascularisation, and stroke. The Grading of Recommendations Assessment, Development and Evaluation (GRADE) framework for prognostic studies was used to determine the level of certainty in the association between complex PCI and the risk of events.
Results: We included 290,039 patients, of whom 94,633 (33%) underwent complex PCI. Compared with non-complex PCI, patients undergoing complex PCI had a higher risk of myocardial infarction (adjusted HR 1.71, 95% credible interval [CrI]: 1.49-1.96), major bleeding (adjusted HR 1.24, 95% CrI: 1.14-1.35), all-cause death (adjusted HR 1.21, 95% CrI: 1.12-1.32), cardiovascular death (adjusted HR 1.29, 95% CrI: 1.15-1.46), stent thrombosis (adjusted HR 1.76, 95% CrI: 1.49-2.14), target lesion or vessel revascularisation (adjusted HR 1.99, 95% CrI: 1.58-2.50), and stroke (adjusted HR 1.21, 95% CrI: 1.03-1.42). The posterior probability of a higher risk associated with complex versus non-complex PCI was >99% for all study outcomes. Except for stroke (which was low certainty), the certainty of evidence was moderate to high for all other outcomes. Secondary analysis, including unadjusted risk estimates, provided consistent results.
Conclusions: Patients undergoing complex PCI have an increased risk of both ischaemic and bleeding events compared with patients undergoing non-complex PCI (PROSPERO: CRD420250656254).
Advances in pharmacological treatments, equipment, and devices have allowed percutaneous coronary intervention (PCI) to be performed in expanding subsets of patients with a large burden of comorbidities and anatomical complexity1. In this context, the term complex PCI has been introduced in the past decade to identify a broad spectrum of interventions with challenging anatomical features in common, including severe calcification, chronic total occlusion, bifurcation lesions, or multivessel treatment23. So far, data on the association between complex PCI and the risk of adverse events have been sparsely reported, and whether complex PCI is independently associated with higher adjusted risks of ischaemic and bleeding events is uncertain. Such information would provide important clinical utility given the implications for procedural strategy and antithrombotic management.
Against this background, we performed a systematic review and Bayesian meta-analysis to evaluate the association between complex PCI and the risk of ischaemic and bleeding outcomes.
Methods
We followed the Prognosis Research Strategy (PROGRESS) group recommendations4567 and the guidance for systematic reviews and meta-analyses of prognostic studies8. The study protocol was registered in the International Prospective Register of Systematic Reviews (PROSPERO: CRD420250656254).
Data sources and searches
Studies were identified by searching MEDLINE, Embase, and PubMed (excluding MEDLINE records) databases from inception to December 2024; Supplementary Table 1 provides the full search strings for each database. We manually screened the reference lists of the eligible articles to check if any relevant articles were missed. The search was restricted to human studies, with no restrictions on language or year of publication.
Study selection
The review question was structured using the Population, Index prognostic factor, Comparator prognostic factors, Outcome, Timing and Setting (PICOTS) framework (Supplementary Table 2)8. Both observational and randomised studies were eligible. Studies were included if complex PCI was defined using more than one of the following procedural criteria: ≥3 stents implanted, ≥3 vessels treated, ≥3 lesions treated, bifurcation lesions requiring implantation of ≥2 stents, total stent length >60 mm, chronic total occlusion PCI, left main PCI, in-stent restenosis, or use of atherectomy. We adopted this definition of complex PCI to account for the heterogeneous distribution of complexity criteria in real-world practice, where individual procedural characteristics frequently coexist. Therefore, including studies that employed a single criterion-based definition would have limited the interpretation of our findings. In case of overlap between studies, we selected the largest study. Two investigators (A.P. Vitale and A. Laino) independently screened titles and abstracts against the eligibility criteria using the Rayyan platform (https://www.rayyan.ai), with potentially eligible studies undergoing full-text assessment. Disagreements were resolved through discussion to achieve consensus.
Outcomes
The two prespecified outcomes of interest were myocardial infarction and major bleeding, given the well-known trade-off between ischaemic and bleeding complications. Additional outcomes included all-cause death, stent thrombosis, cardiovascular (or cardiac) death, target lesion or vessel revascularisation, and stroke. Myocardial infarction was categorised as spontaneous or periprocedural. When available, risk estimates were preferentially extracted according to the Fourth Universal Definition of Myocardial Infarction; otherwise, we extracted the study-level definition (Supplementary Table 3). For major bleeding, we preferentially extracted risk estimates for the composite of Type 3 or 5 bleeding, according to the Bleeding Academic Research Consortium (BARC) scale9. If unavailable, we sequentially extracted risk estimates for Thrombolysis in Myocardial Infarction (TIMI) major bleeding, Global Utilization of Streptokinase and Tissue plasminogen activator for Occluded coronary arteries (GUSTO) moderate or severe bleeding, or Randomized Evaluation in PCI Linking Angiomax to Reduced Clinical Events (REPLACE-2) major bleeding. Further details on outcome definitions are reported in Supplementary Table 3. Post hoc outcomes were a device-oriented composite endpoint (DOCE), as defined in each included study, spontaneous myocardial infarction, and target vessel myocardial infarction.
Data extraction and quality assessment
Data extraction was performed using a predesigned data extraction sheet, developed according to the modified version of the CHecklist for critical Appraisal and data extraction for systematic Reviews of prediction Modelling Studies (CHARMS) for prognostic factors810. The definition of complex PCI for each included study is detailed in Supplementary Table 4. Two reviewers independently collected hazard ratios (HRs) for the outcomes of interest and assessed the risk of bias of included studies, using the Quality in Prognosis Studies (QUIPS) tool11. This tool includes six potential domains of bias: study participation, study attrition, prognostic factor measurement, outcome measurement, confounding measurement, and statistical analysis and reporting. Disagreements were resolved through discussion until consensus was reached.
Data synthesis and statistical analysis
The primary analysis was conducted in studies that provided adjusted risk estimates (the covariates used by each study are listed in Supplementary Table 5). Secondary analysis using unadjusted risk estimates was also performed but reported separately. All analyses were carried out using random-effects models within a Bayesian framework. This approach was chosen because it improves clinicians’ interpretation of the meta-analysis results and the evaluation of between-study heterogeneity12. Specifically, we selected vague priors: for the log of the HR, we used a normally distributed prior with a mean of 0 and a standard deviation of 2; for the heterogeneity parameter (τ), we used a half-normal prior with a scale of 0.5. Risk estimates were summarised using pooled HRs with 95% credible intervals (CrIs). In addition, we reported the posterior probabilities of complex PCI being associated with a higher risk of an event compared with non-complex PCI (i.e., the probability that the HR was >1). As a sensitivity analysis, we used non-informative priors given the anticipated high number of studies included in the quantitative meta-analysis, as well as a frequentist random-effects meta-analysis created by pooling HRs, with a generic inverse-variance model (using the Hunter-Schmidt estimates of the between-study variance)13. We evaluated heterogeneity across studies with τ and I2 statistics. Small-study effects were evaluated by visual assessment of the contour-enhanced funnel plots and using the formal Egger’s regression test14.
We investigated potential sources of heterogeneity in the primary outcomes by post hoc meta-regression analyses, including information on baseline characteristics, known predictors of ischaemia15, known predictors of bleeding15, antithrombotic therapies, complex PCI components, and year of study publication. In addition, sensitivity analyses were performed for both primary (prespecified) and secondary (post hoc) outcomes by separately evaluating observational versus randomised studies, as well as studies enrolling patients with acute coronary syndrome and those including patients with chronic coronary syndrome.
Two reviewers evaluated the overall certainty in pooled estimates using the Grading of Recommendations Assessment, Development and Evaluation (GRADE) approach. These estimates were categorised into one of four levels of certainty: high, moderate, low, or very low. In accordance with GRADE guidance for prognostic studies, cohort data are considered high-certainty evidence16. All statistical analyses were performed using R software (R Foundation for Statistical Computing; using the bayesmeta package)17.
Results
Study selection and patient population
From 13,616 articles, 460 were identified as potentially eligible records after the initial screening (Supplementary Figure 1). Of these, 374 were excluded due to incompatible populations, 40 for incomplete data, 7 for missing outcomes, and 3 for overlapping populations. As a result, we included 36 studies with 290,039 patients, of whom 94,633 (33%) underwent complex PCI and 195,406 (67%) underwent non-complex PCI (Table 1). The criteria used for defining complex PCI are shown in Table 1. Supplementary Table 6-Supplementary Table 7-Supplementary Table 8-Supplementary Table 9-Supplementary Table 10-Supplementary Table 11 summarise the patient-level and study-level characteristics. The median age of complex PCI patients was 65.5 years (interquartile range [IQR] 63.9-68.9). The median proportion of patients with diabetes in the complex PCI group was 35% (IQR 32.8-43.0). Acute coronary syndrome was the indication for PCI in 51.95% (IQR 35.5-70.0) of complex PCI patients. From the 36 included studies, a total of 27 studies reporting adjusted HRs were included in the primary analysis, while 27 studies, which provided unadjusted risk estimates, were included in the secondary analysis. The median follow-up duration was 1 year. The studies were published between 2016 and 2024, and most studies were observational (24 out of 36). Overall, the antithrombotic therapies used during and after PCI were comparable between patients undergoing complex PCI versus non-complex PCI.
Table 1. Characteristics of the population included in the meta-analysis.
| Population | Value |
|---|---|
| Sample size | 290,039 |
| Complex PCI | 28.3% |
| Females in complex PCI group | 24.1% |
| Diabetic patients in complex PCI group | 35% |
| ACS in complex PCI group | 51.9% |
| Median follow-up, years | 1 |
| Components of complex PCI | |
| ≥3 stents implanted | 16.05 (9.23-21.35) |
| ≥3 vessels treated | 3.14 (2.35-5.45) |
| ≥3 lesions treated | 7.5 (5.2-10.2) |
| Bifurcation with ≥2 stents implanted | 5.6 (3.1-9.5) |
| Total stent length >60 mm implanted | 14.85 (8.8-19.95) |
| Chronic total occlusion PCI | 5 (3.98-8.92) |
| Saphenous vein graft PCI | 5 (5.1-5.2) |
| Left main coronary artery PCI | 4 (3.5-6.4) |
| In-stent restenosis | 2.8 (2.0-3.6) |
| Rotational atherectomy | 8.2 (2.1-8.4) |
| Population | No. of studies |
| Continent of study | |
| Europe | 11 (30) |
| North America | 9 (25) |
| Asia/Oceania | 16 (45) |
| Africa | 0 (0) |
| Study design | |
| Observational | 24 (67) |
| Randomised | 12 (33) |
| Multicentre study | |
| No | 9 (25) |
| Yes | 27 (75) |
| Presentation at index PCI | |
| Acute coronary syndrome | 7 (20) |
| Chronic coronary syndrome | 4 (11) |
| Acute or chronic coronary syndrome | 25 (69) |
| Risk estimates | |
| Adjusted | 27 (75) |
| Unadjusted | 9 (25) |
| Categorical variables are expressed as median percentages. Other data are given as n, n (%), or median (IQR). ACS: acute coronary syndrome; IQR: interquartile range; PCI: percutaneous coronary intervention | |
Risk of bias
Supplementary Table 12 presents the risk-of-bias assessment. Most of the studies were at low to moderate risk of bias. All unadjusted studies were deemed at high risk of bias due to the lack of adjustments for other prognostic factors.
Primary analysis (adjusted risk estimates)
Out of the 222,001 included participants, 78,520 (35%) underwent complex PCI.
Myocardial infarction
A total of 19 studies, including 150,213 participants, of whom 57,626 underwent complex PCI, contributed to the analysis of myocardial infarction. Using a Bayesian random-effects model with vague priors, patients undergoing complex PCI had a higher risk of myocardial infarction (adjusted HR 1.71, 95% CrI: 1.49-1.96; moderate to substantial heterogeneity [τ=0.24, I2=70%]; high certainty). We found a 99.9% posterior probability that complex PCI was associated with an increased risk of myocardial infarction (Figure 1). These findings were consistent when using uninformative priors and a frequentist approach (Figure 1A).
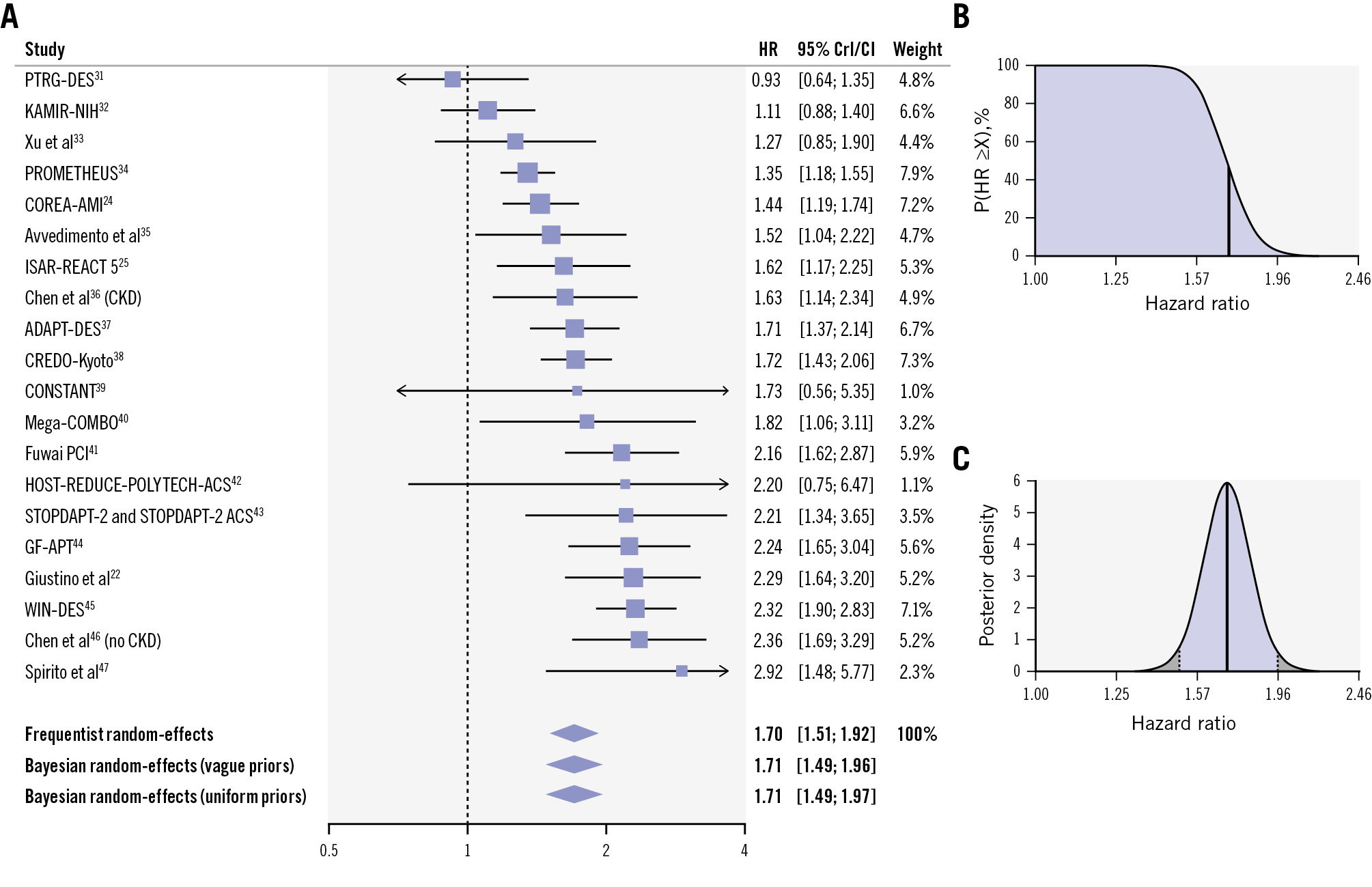
Figure 1. Adjusted risk of myocardial infarction in patients undergoing complex versus non-complex PCI. A) Forest plot of adjusted analysis for myocardial infarction. B) Cumulative posterior distribution of the estimated adjusted hazard ratio for myocardial infarction. On the y-axis, there is the probability (P) that the hazard ratio is greater than or equal to the value on the x-axis (X). The bold vertical line indicates the median value. The blue area is indicative of a good prognostic ability of complex PCI criteria (probability that the hazard ratio is greater than or equal to 1). C) Full posterior distribution of the estimated adjusted hazard ratio for myocardial infarction. The bold vertical line indicates the median value, and the area highlighted in blue indicates the percentile-based 95% credible interval. CI: confidence interval; CKD: chronic kidney disease; CrI: credible interval; HR: hazard ratio; PCI: percutaneous coronary intervention
Major bleeding
Data from 13 studies, comprising 101,096 patients, of whom 33,740 underwent complex PCI, were analysed. Using a Bayesian random-effects model with vague priors, patients undergoing complex PCI had a higher risk of major bleeding compared with those undergoing non-complex PCI (adjusted HR 1.24, 95% CrI: 1.14-1.35; low heterogeneity [τ=0.08, I2=24%]; high certainty). There was a 99.9% posterior probability that complex PCI was associated with an increased risk of major bleeding (Figure 2). Findings were consistent when using uninformative priors and a frequentist model (Figure 2A).
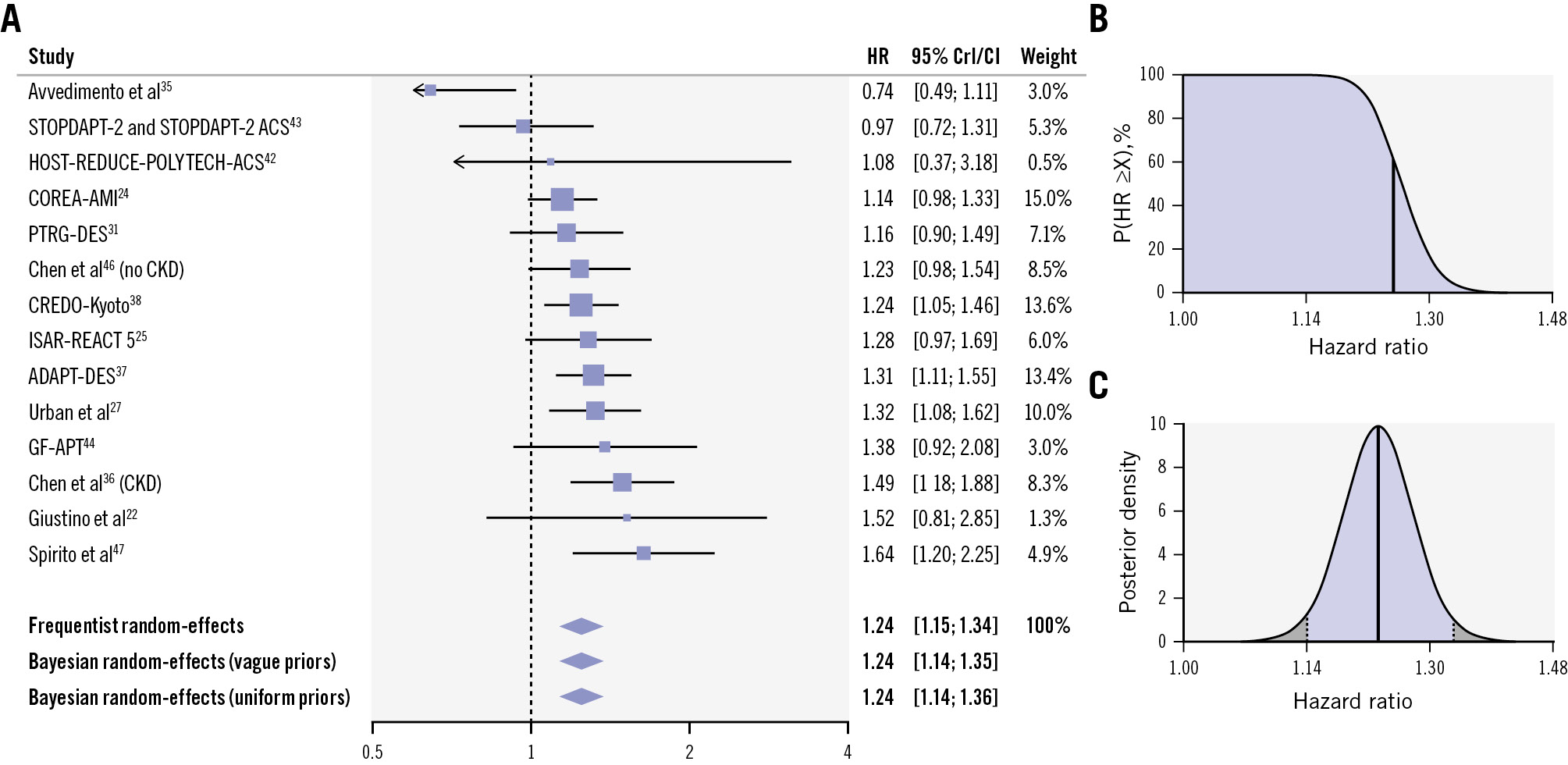
Figure 2. Adjusted risk of major bleeding in patients undergoing complex versus non-complex PCI. A) Forest plot of adjusted analysis for major bleeding. B) Cumulative posterior distribution of the estimated adjusted hazard ratio for major bleeding. On the y-axis, there is the probability (P) that the hazard ratio is greater than or equal to the value on the x-axis (X). The bold vertical line indicates the median value. The blue area is indicative of a good prognostic ability of complex PCI criteria (probability that the hazard ratio is greater than or equal to 1). C) Full posterior distribution of the estimated adjusted hazard ratio for major bleeding. The bold vertical line indicates the median value, and the area highlighted in blue indicates the percentile-based 95% credible interval. CI: confidence interval; CKD: chronic kidney disease; CrI: credible interval; HR: hazard ratio; PCI: percutaneous coronary intervention
All-cause death
A total of 21 studies, including 150,744 patients, of whom 56,746 underwent complex PCI, contributed to the analysis of all-cause death. The risk of all-cause death was higher in patients undergoing complex versus non-complex PCI (adjusted HR 1.21, 95% CrI: 1.12-1.32; moderate heterogeneity [τ=0.12, I2=43%]; moderate certainty), with a 99.9% posterior probability (Figure 3, Supplementary Figure 2). Results were confirmed using uninformative priors and a frequentist model (Figure 3).
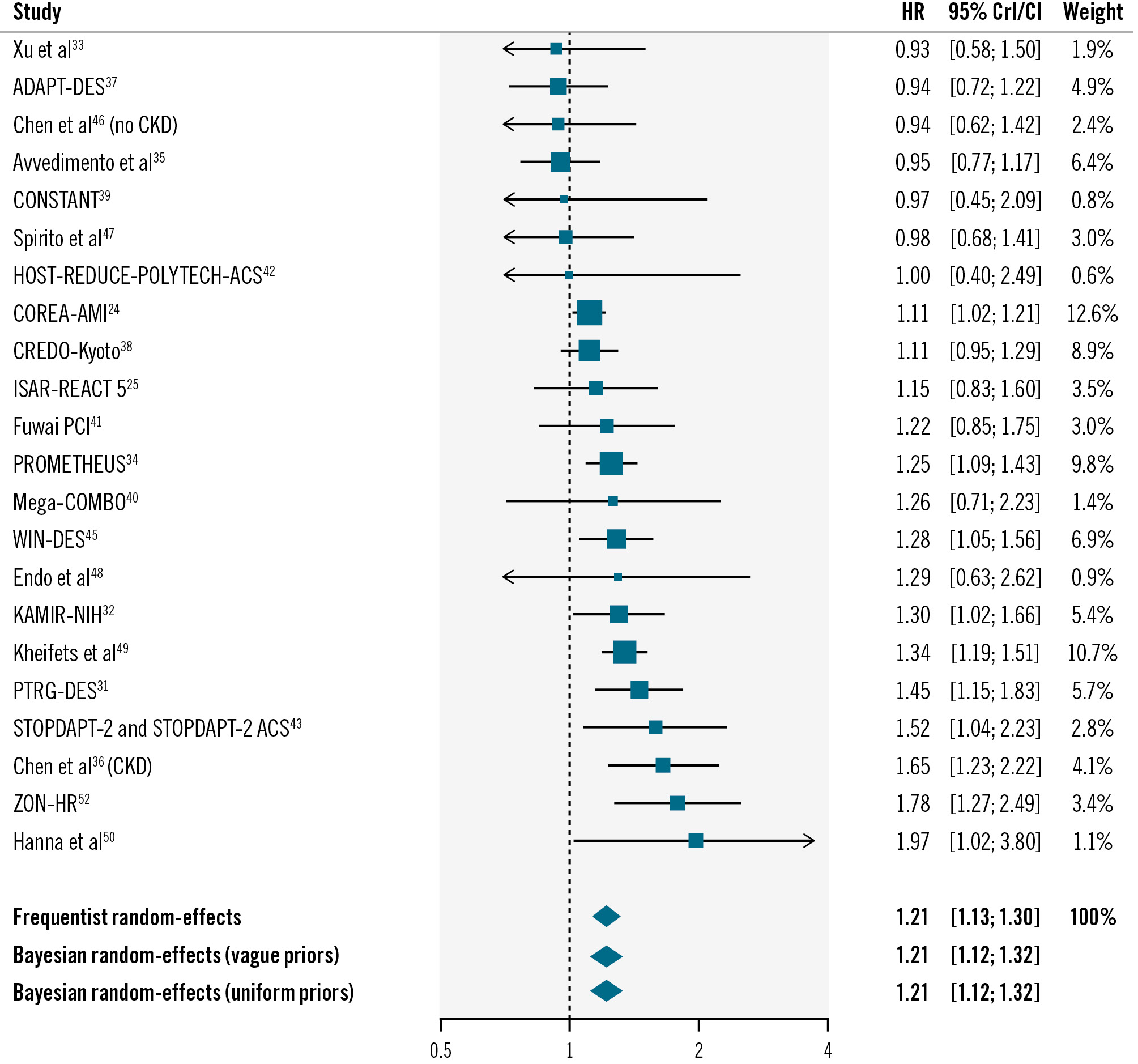
Figure 3. Adjusted risk of all-cause death in patients undergoing complex versus non-complex PCI. CI: confidence interval; CKD: chronic kidney disease; CrI: credible interval; HR: hazard ratio; PCI: percutaneous coronary intervention
Other outcomes
A total of 16 studies, enrolling 144,138 participants (50,168 underwent complex PCI), provided data regarding stent thrombosis. Complex PCI was associated with a higher risk of stent thrombosis (adjusted HR 1.71, 95% CrI: 1.45-2.04, posterior probability 99.9%; low heterogeneity [τ=0.10, I2=24%]; high certainty) (Figure 4, Supplementary Figure 3). Fifteen studies, including 127,100 patients, contributed to the analysis of cardiovascular death. Complex PCI was associated with a higher risk of cardiovascular death (adjusted HR 1.29, 95% CrI: 1.15-1.46, posterior probability 99.9%; low heterogeneity [τ=0.12, I2=24%]; high certainty) (Supplementary Figure 4, Supplementary Figure 5). Sixteen studies with a total of 125,057 participants provided data on target lesion or vessel revascularisation. Complex PCI showed an increased risk of target lesion or vessel revascularisation (adjusted HR 1.99, 95% CrI: 1.58-2.49, posterior probability 99.9%; high heterogeneity [τ=0.39, I2=92%]; moderate certainty) (Supplementary Figure 6, Supplementary Figure 7). Stroke was evaluated in 10 studies with 69,091 participants. Patients undergoing complex PCI showed an increased risk of stroke (adjusted HR 1.21, 95% CrI: 1.03-1.42, posterior probability 98.4%; moderate heterogeneity [τ=0.08, I2=30%]; low certainty) (Supplementary Figure 8, Supplementary Figure 9).
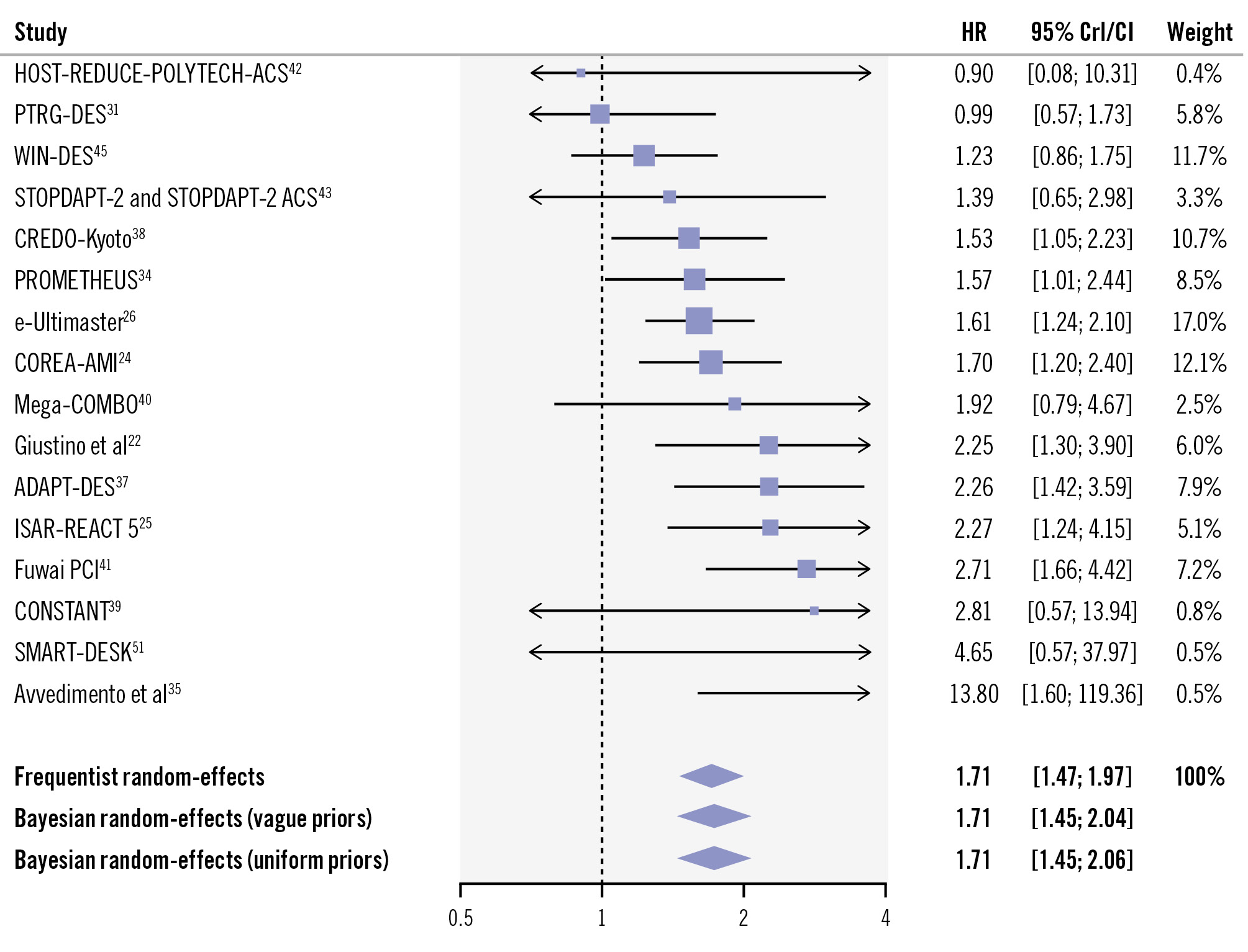
Figure 4. Adjusted risk of stent thrombosis in patients undergoing complex versus non-complex PCI. CI: confidence interval; CrI: credible interval; HR: hazard ratio: PCI: percutaneous coronary intervention
Post hoc outcomes
Patients undergoing complex PCI had a greater risk of the DOCE, spontaneous myocardial infarction, and target vessel myocardial infarction compared with patients undergoing non-complex PCI (Supplementary Appendix 1-Supplementary Appendix 2-Supplementary Appendix 3).
Secondary analysis (unadjusted risk estimates)
We analysed 186,788 participants from 27 studies, including 56,908 patients undergoing complex PCI. Principal results are shown in Supplementary Figure 10-Supplementary Figure 11-Supplementary Figure 12-Supplementary Figure 13-Supplementary Figure 15-Supplementary Figure 16.
Supplementary Table 13 summarises the results of the Bayesian analysis. Overall, the unadjusted and adjusted analyses were largely consistent (Central illustration).
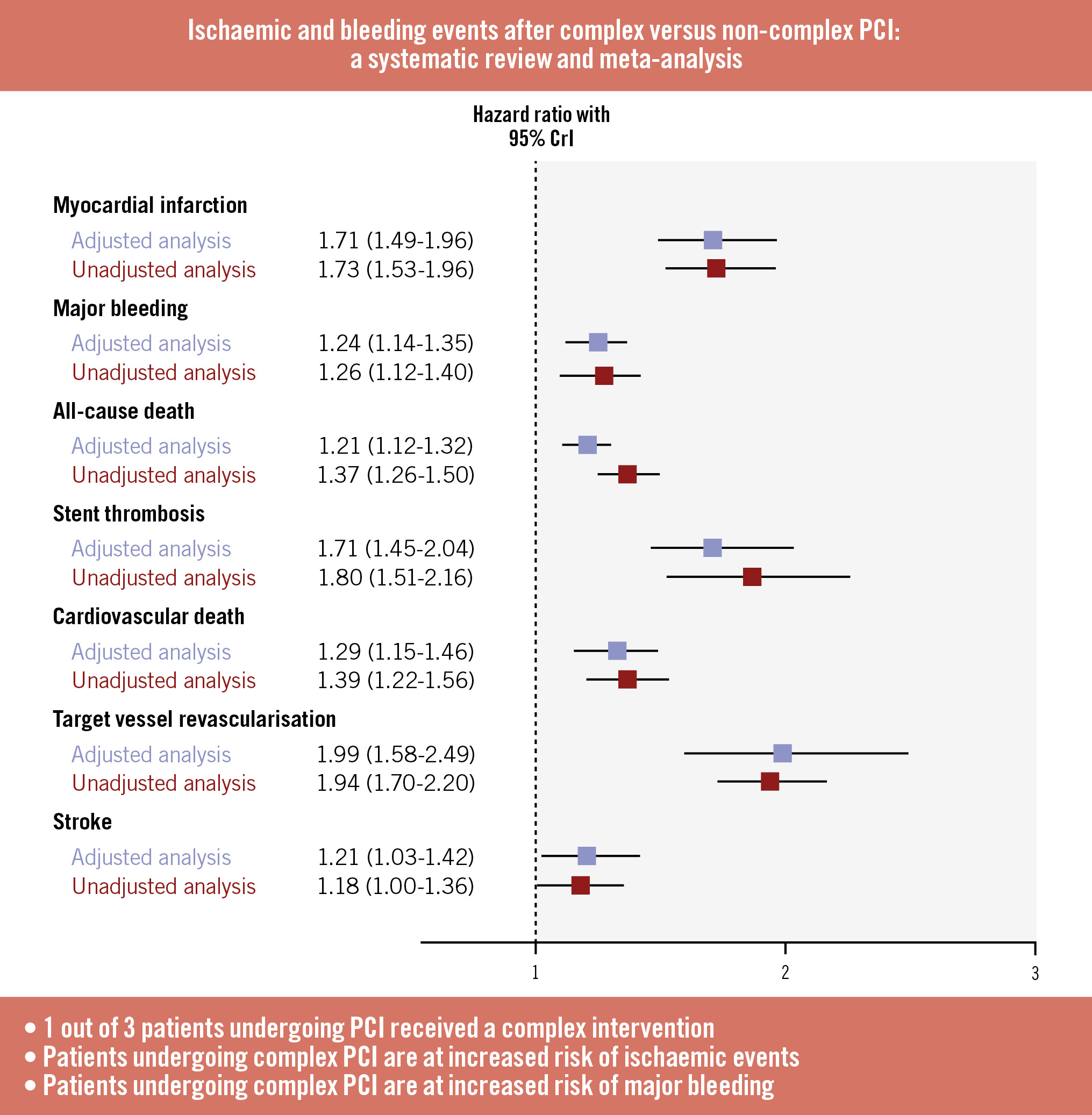
Central illustration. Summary forest plot for each outcome included in the adjusted and unadjusted analyses. CrI: credible interval; PCI: percutaneous coronary intervention
Meta-regression analysis
Meta-regression analysis revealed that ethnicity, the proportion of patients with acute coronary syndrome, prior myocardial infarction, prior cerebrovascular accident, elderly status, and the proportion of patients with a stent >60 mm implanted were the major sources of the heterogeneity observed for the primary outcomes (Supplementary Table 14-Supplementary Table 15-Supplementary Table 16-Supplementary Table 17-Supplementary Table 18-Supplementary Table 19).
Subgroup analysis
Results remained consistent when data were restricted to observational studies, randomised trials, studies including patients with chronic coronary syndrome, and studies including patients with acute coronary syndrome (Supplementary Table 20).
Reporting bias and level of evidence
No publication bias was detected based on the funnel plots or Egger’s tests (Supplementary Figure 17-Supplementary Figure 18-Supplementary Figure 19-Supplementary Figure 20-Supplementary Figure 21-Supplementary Figure 22-Supplementary Figure 23). The GRADE assessment rated the evidence as high or moderate certainty, except for stroke (which was low certainty) (Supplementary Table 21).
Discussion
In the present systematic review and meta-analysis of 290,039 patients from 36 studies, we evaluated the association between complex PCI and several clinically relevant outcomes. Our main findings are as follows: (1) 1 out of 3 patients treated with percutaneous revascularisation underwent complex PCI; (2) complex PCI was associated with a ~70% relative increase in the adjusted risk of myocardial infarction, with ~99% posterior probability and a high certainty of evidence; (3) complex PCI was associated with a ~25% relative increase in the risk of major bleeding, with ~99% posterior probability and a high certainty of evidence; (4) compared with non-complex PCI, patients undergoing complex PCI had a ~20% higher risk of all-cause death (moderate certainty), ~70% higher risk of stent thrombosis (high certainty), ~100% higher risk of target lesion or vessel revascularisation (moderate certainty), and ~20% higher risk of stroke (low certainty).
The present analysis is the first systematic, comprehensive study mapping the prognostic impact of complex PCI on major cardiovascular outcomes. Coronary artery disease (CAD) complexity and high-risk patient characteristics are well-known predictors of adverse events. Prior large-scale analyses were primarily focused on the predictive value of CAD complexity by relying on anatomical factors18 or combining clinical and procedural factors19. However, the definition of complex PCI deviates from the concepts related to CAD complexity or high-risk patient profiles. While the latter can be categorised as patient- or lesion-focused, complex PCI includes a broader range of lesion and procedural characteristics20 and, in the past few years, has emerged as a key subgroup of interest in randomised trials across several domains. Despite the lack of universally agreed criteria for complex PCI, the definitions of complex PCI were largely consistent across the included studies.
The impact of complex PCI on clinical outcomes has been investigated in prior analyses; however, our study is the first attempt to quantitatively define the magnitude of this association. We found that patients undergoing complex PCI had a ~70% relative increase in the risk of myocardial infarction, a finding that remained largely unchanged in the unadjusted analyses. A similar increase in the order of 70-100% was also found for other ischaemic events, such as stent thrombosis and target lesion or vessel revascularisation. These findings align well with prior studies showing an elevated ischaemic risk in patients treated with complex PCI2122. The underlying reasons for the heightened ischaemic risk after complex PCI are mechanistically plausible and somewhat expected. Patients undergoing complex PCI have a more severe and advanced atherosclerotic burden, along with greater plaque instability. In addition, the complexity of intervened lesions (e.g., long lesions, bifurcations, severe calcifications) often requires multiple and overlapping stents, potentially leading to delayed endothelisation, stent malapposition, and incomplete lesion coverage, with a higher risk of vessel injury or dissection, all predisposing to stent thrombosis or periprocedural myocardial infarction2324. Moreover, multivessel CAD is often associated with incomplete myocardial revascularisation, with the corollary effect of a greater burden of untreated residual coronary lesions that predispose patients to future acute events.
Although the magnitude of association was lower compared with myocardial infarction, complex PCI was associated with an approximately 20% increased risk of major bleeding. Prior evidence regarding the impact of complex PCI on bleeding events was inconclusive. An analysis from Intracoronary Stenting and Antithrombotic Regimen: Rapid Early Action for Coronary Treatment (ISAR-REACT) 5 and a pooled analysis of six trials did not find any difference in the risk of major bleeding between complex and non-complex PCI22232425. Conversely, in a large, unselected cohort of patients undergoing PCI, both ischaemic and bleeding events were increased among patients undergoing complex, in comparison with non-complex, PCI24. Mechanistically, the increased risk of bleeding could be explained by the more frequent use of femoral access, larger sheaths, and more potent antithrombotic therapy during complex PCI. Moreover, several patient-related factors are more commonly found among patients undergoing complex PCI, including advanced age, comorbidities, chronic oral anticoagulation, anaemia, gastrointestinal disease, or malignancy26. Interestingly, many of these features overlap with high bleeding risk (HBR) criteria, such as older age, renal dysfunction, anaemia, and chronic anticoagulation – further supporting the relationship between complex PCI and HBR status27.
Historically, complex PCI has been recognised as a high ischaemic risk condition, with bleeding risk often overlooked. Conversely, our analysis supports the concept that complex PCI confers a dual hazard with increases in both ischaemic and bleeding risks. This highlights the need to pursue bleeding-avoidance strategies also in patients undergoing complex PCI. During the periprocedural period, the preferential use of radial access, the implementation of ultrasound-guided femoral access, and careful selection of anticoagulant therapy may mitigate the bleeding risk28. On the other hand, the excess of ischaemic events, including stent thrombosis, repeat revascularisation, and myocardial infarction, among patients undergoing complex PCI deserves careful attention when these procedures are performed. In this context, the extensive use of intracoronary imaging, which has proved to be particularly useful in reducing ischaemic recurrences29, may be helpful for complex PCI procedures. With respect to antithrombotic strategies, our results, which align with prior studies2130, provide further evidence supporting the prioritisation of bleeding over ischaemic risk assessment when selecting the appropriate dual antiplatelet therapy regimen for CAD patients. Therefore, long-term antiplatelet strategies, including de-escalation or P2Y12 inhibitor monotherapy, should also be considered in this population given their similar efficacy and improved safety – also in the context of complex PCI221. However, it should be acknowledged that our findings were influenced by the high prevalence of patients with HBR undergoing complex PCI. Unfortunately, subgroup analyses comparing HBR versus non-HBR patients were not feasible. Therefore, our analyses cannot determine whether such risk stratification may identify a subset of complex PCI patients who could benefit from more intensive dual antiplatelet therapy.
Limitations
Our study has several limitations. First, we observed a moderate to high degree of between-study heterogeneity in some analyses. This finding is common in meta-analyses of prognostic studies and may reflect differences in the definition of complex PCI, outcome definitions, covariates included in adjustment models, and inconsistencies in the definition and measurement of these covariates across studies. In this context, our results were generally consistent across multiple subgroup and meta-regression analyses. However, the use of study-level rather than individual-level data precluded a comprehensive assessment of the influence of these potential modifiers on the observed treatment effects. Second, we were unable to provide separate analyses for early versus late outcomes. This analysis would have been important to disentangle the periprocedural risk associated with complex PCI from the long-term risk. Third, our study demonstrated the association between complex PCI and increased risks of both ischaemic and bleeding events. Although these findings are informative for risk stratification, they do not directly guide the selection of antithrombotic treatment strategies after PCI. These associations should be interpreted with caution and ideally confirmed in adequately powered randomised clinical trials that are specifically designed to evaluate antithrombotic strategies stratified by PCI complexity. Fourth, several included studies were not specifically designed to enrol patients with complex CAD. Therefore, highly complex forms or subsets may have been underrepresented or excluded. Fifth, so far, there is no universal definition of complex PCI. As a result, different subtypes of complex PCI, which may carry distinct prognostic implications, are often grouped under the same diagnostic category. Currently, data are not systematically reported in a way that allows detection of these differences, thereby limiting the ability to explore the differential prognostic implications of specific complex PCI patterns. This limitation may have introduced a degree of bias into our analyses and contributed to residual heterogeneity that could not be adequately accounted for. Finally, although our meta-regression analyses did not identify any interaction between antithrombotic therapies and the observed treatment effects, some limitations should be taken into consideration. These analyses, which were powered only when a high number of studies were included, were conducted on a post hoc basis. In addition, information regarding antithrombotic therapy was inconsistently reported across studies. As a result, the lack of relationship between antithrombotic therapy and treatment effect should be interpreted with caution.
Conclusions
In conclusion, our systematic review and meta-analysis, which included 290,039 patients from 36 studies, found that complex PCI was associated with a higher risk of ischaemic events, including myocardial infarction, stent thrombosis, and target lesion or vessel revascularisation, than non-complex PCI. Additionally, complex PCI carried a higher risk of major bleeding. While the magnitude and certainty of these risks vary, the associations were consistent across both adjusted and unadjusted analyses. Collectively, these findings underscore the increased risks of complex PCI in terms of both ischaemic and bleeding complications.
Impact on daily practice
Complex percutaneous coronary intervention (PCI) refers to a broad spectrum of interventions characterised by challenging anatomical features, such as severe calcification, chronic total occlusion, bifurcation lesions, or multivessel treatment. Data from several studies indicate a higher baseline risk profile of patients undergoing complex PCI. Compared with non-complex PCI, complex PCI was associated with a significantly higher risk of ischaemic events, including myocardial infarction, stent thrombosis, and target lesion or vessel revascularisation, as well as an increased risk of bleeding events. Complex PCI represents a specific subset of patients requiring tailored strategies aimed at reducing both ischaemic and bleeding events.
Funding
This work is supported by a research grant from the Italian Ministry of Education (PRIN PNRR 2022497RZ4 to R. Piccolo). There was no industry involvement in the design, analysis, or funding of this study.
Conflict of interest statement
G. Esposito reports personal fees from Amgen, Boehringer Ingelheim, Edwards Lifesciences, and Sanofi, outside the submitted work; and research grants to the institution from Boston Scientific and Medtronic. The other authors have no conflicts of interest to declare.
Supplementary data
To read the full content of this article, please download the PDF.

